
HS Dosing COSENTYX® (secukinumab)
4.6
(723)
Escribir comentario
Más
€ 10.99
En stock
Descripción

European Commission Approves Secukinumab for Moderate-to-Severe Hidradenitis Suppurativa - LiVDerm

FDA approves Novartis' Cosentyx

WO2019097493A1 - Treating hidradenitis suppurativa with il-17 antagonists - Google Patents

COSENTYX® (secukinumab) for US HCP

COSENTYX Dosage & Rx Info

FDA Approves Secukinumab for Hidradenitis Suppurativa
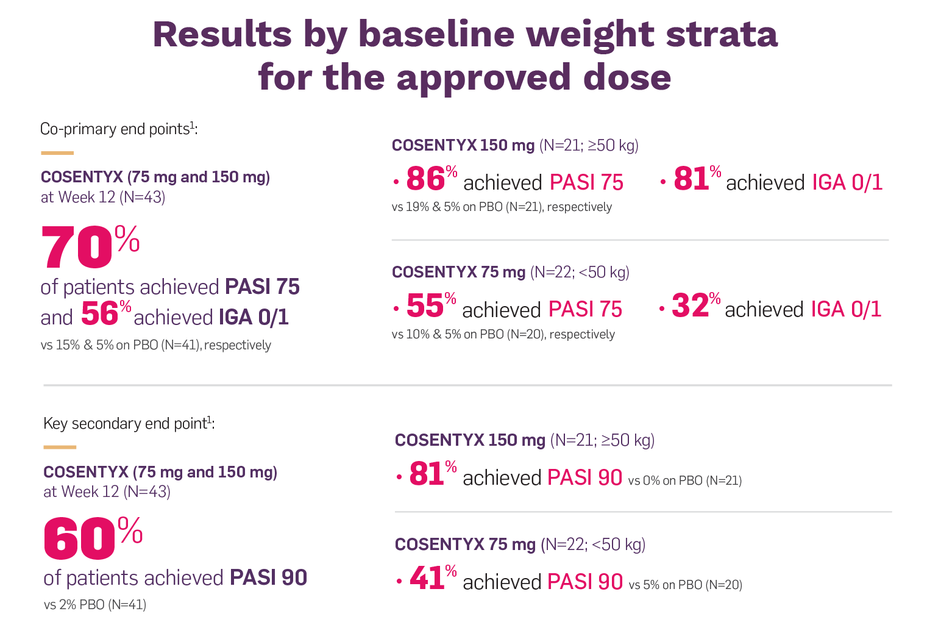
Pediatric PsO Treatment Information, COSENTYX® (secukinumab)

HS Dosing COSENTYX® (secukinumab)

Cosentyx - NPS MedicineWise

US approval for fourth Cosentyx indication

FDA Approves Novartis' Cosentyx® for Hidradenitis Suppurativa American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology
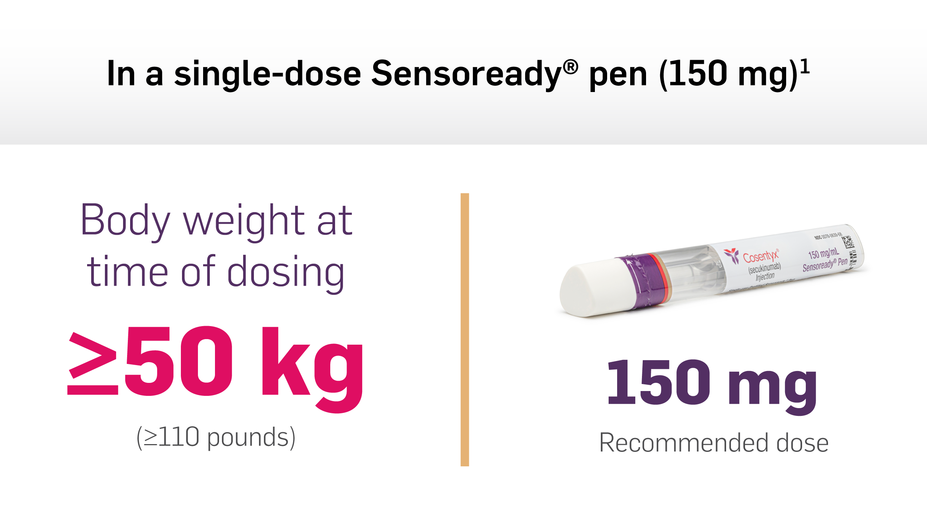
Dosing & Devices, COSENTYX® (secukinumab)
También te puede interesar





:format(jpg)/f.elconfidencial.com%2Foriginal%2F39c%2Fe3b%2Fb03%2F39ce3bb03350b9756281d23bdcb4a847.jpg)



